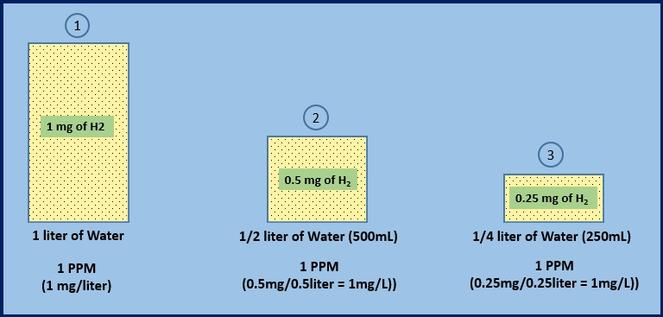
Concentration of solutions Parts per million (ppm) This is a way of expressing very dilute concentrations of substances. Just as per cent means out of. - ppt download

SOLVED: Percent, by definition, is parts per hundred (%). For very dilute solutions, chemists will often use parts per thousand or parts per million ( ppm) or parts per billion (ppb). Calculate the

Parts per Million Concentration. Parts per Million (ppm) is the ratio of the number of grams of solute for every one million grams of solution. parts. - ppt download