Derive a formula for radius of the stable orbit of hydrogen atom on the basis of Bohr model. Prove that in hydrogen - Sarthaks eConnect | Largest Online Education Community
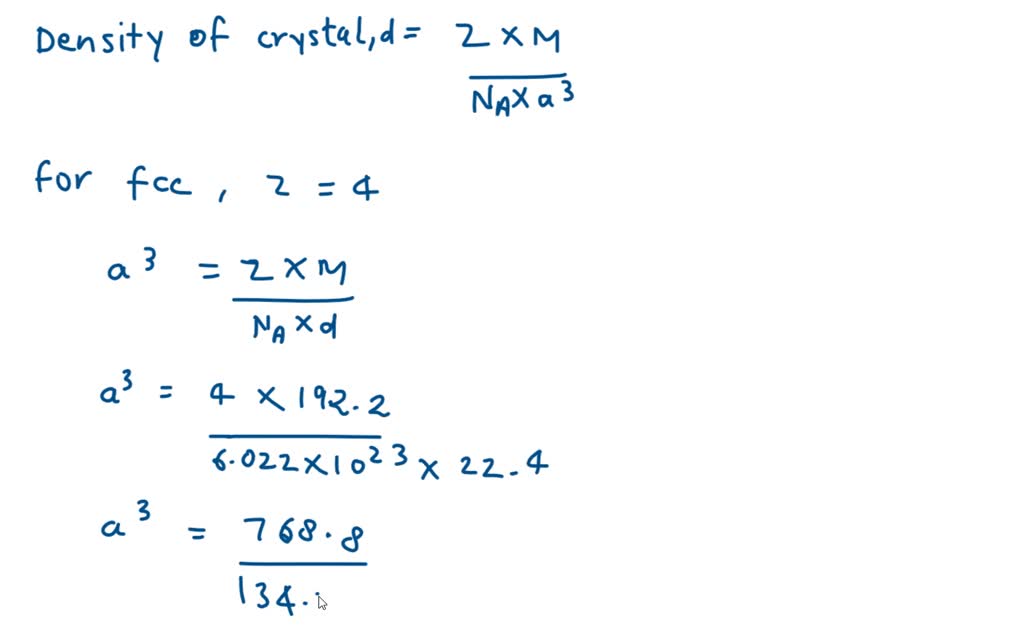
SOLVED: Calculate the radius of an iridium atom, given that ir has an fcc crystal structure, a density of 22.4 g/cm3 , and an atomic weight of 192.2 g/mol.
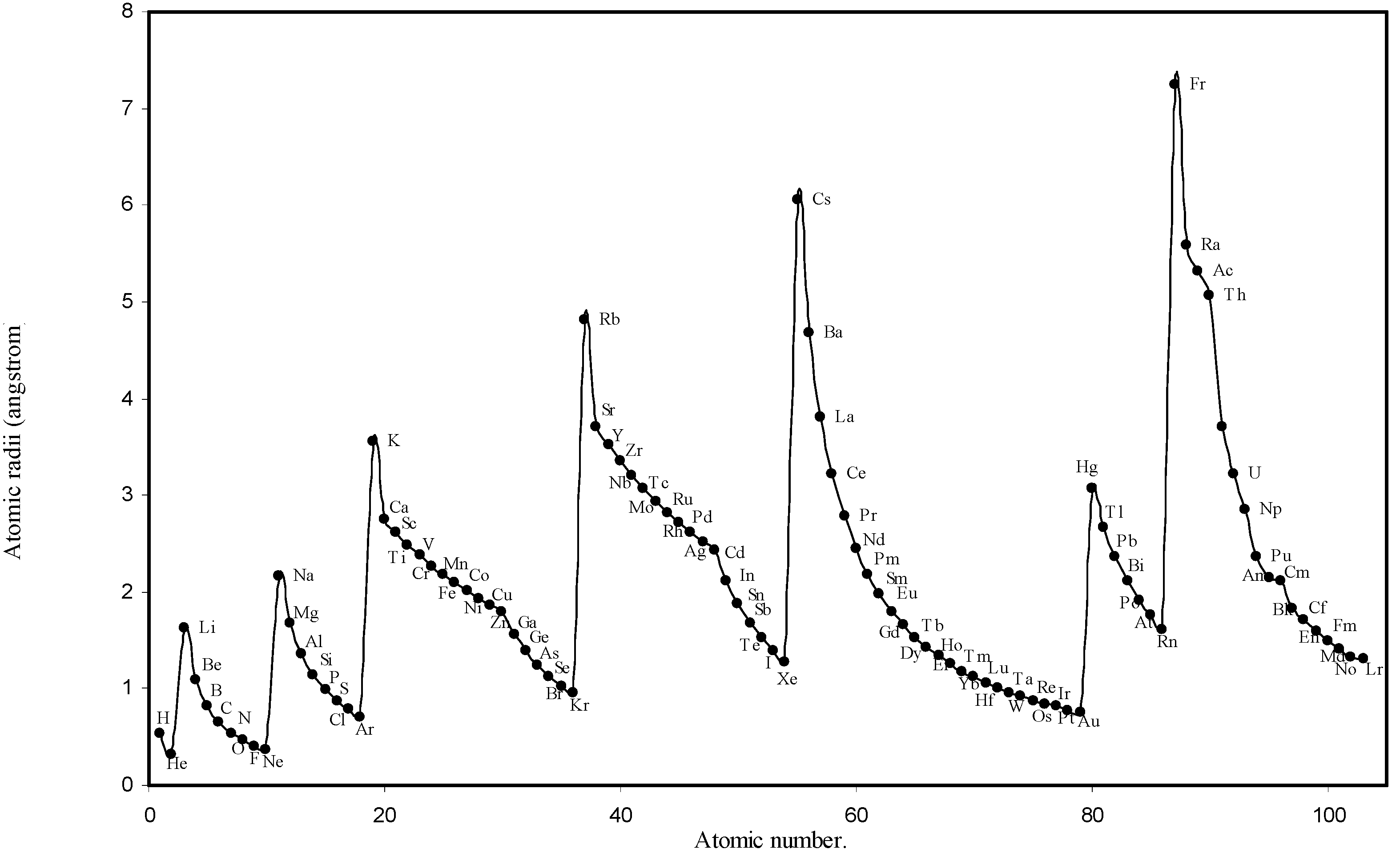
IJMS | Free Full-Text | Theoretical Calculation of Absolute Radii of Atoms and Ions. Part 1. The Atomic Radii

Niobium crystallises in body - centred cubic structure. If density is 8.55 g cm^-3 , calculate atomic radius of niobium using its atomic mass 93 U .

Atomic radius of `Li is 1.23 Å` and ionic radius of `Li^(+)` is `0.76 Å`. Calculate the percentage - YouTube
Derive a formula for radius of the stable orbit of hydrogen atom on the basis of Bohr model. Prove that in hydrogen - Sarthaks eConnect | Largest Online Education Community

Calculate atomic radius using diatomic molecules: oxygen, hydrogen, wall mural • murals structure, molecular, bromine | myloview.com
What is the formula to calculate the radius of an orbit of the atom and velocity of the specific shell of the atom.

Atomic radius is of order 10^-8 cm and nuclear radius is of order 10^-13. calculate what fraction of atom is occupied by nucleus? | Socratic